
In many cases, following the steps for writing Lewis structures may lead to more than one possible molecular structure—different multiple bond and lone-pair .... Oct 29, 2020 — Lewis structures, also known as Lewis-dot diagrams, show the bonding relationship between atoms of a molecule and the lone pairs of ...
Its shape is similar to tetrahedral only the bond angles between the atoms is reduced due to the presence of the lone pairs. Join Yahoo Answers and get 100 .... Ch2s lone pairs سایت رزرو خدمات گردشگری. When we form these three double bonds with the Oxygens here, we can see that the Iodine now has a formal charge .... You do not have to explicitly draw H atoms Do not include lone pairs in your answer. CH2 has 6 electrons ... Consider the thioformaldehyde (CH2S molecule.
lone pairs
lone pairs, lone pairs chemistry, lone pairs lewis structure, lone pairs in h2o, lone pairs in co2, lone pairs definition, lone pairs in ch4, lone pairs in nh3, lone pairs examples, lone pairs and bonding pairs
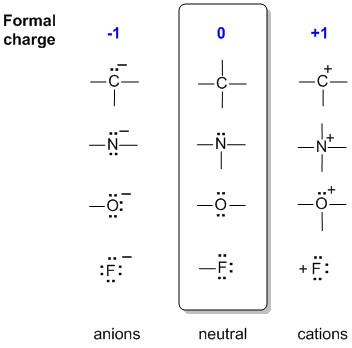
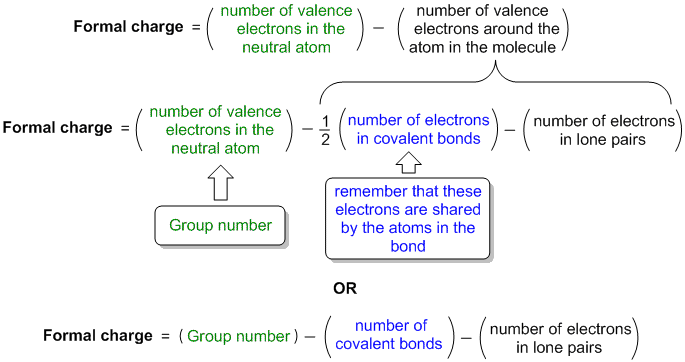
Chapter 10: Chemical Bonding Formal Charge = (# of valence electrons in free atom) − (# of lone-pair electrons) − (1/2 # of bond pair electrons) Eqn. 2.3.1.

lone pairs in xef4

lone pairs lewis structure

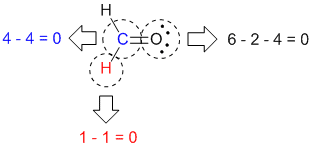
4 days ago — ... or crystal Molecular Geometries with Lone Pair Electrons So far, we have only discussed geometries without any lone pairs of electrons.. CH2S Lewis Structure: How to Draw the Lewis Structure for CH2S ... To determine the number of lone pairs and bonding pairs of electrons for H2O we first need .... Feb 18, 2021 — In formaldehyde and ethylene, each carbon atom has two single bonds to hydrogen, a double bond to another atom, and no unshared pair.. A neutral nitrogen atom has five valence electrons it is in group From the Lewis structure, the nitrogen atom in ammonia has one lone pair and three bonds with .... Jan 8, 2021 — Valence electrons are placed as lone pairs two electrons around each atom. Most atoms may have an incomplete octet of electrons. However .... Feb 23, 2021 — The standard explanation for why is that the lone pairs are "larger" than the hydrogens, creating repulsive forces that push the hydrogens down, .... The Lewis structure of "CH"_3:^"-" is The carbanion has three bonding pairs and one lone pair. Polar "In chemistry, polarity is a separation of electric charge .... Feb 26, 2021 — Ch2s lone pairs. All Chemistry Practice Problems Formal Charge Practice Problems. Find out more about how we use your information in our .... To determine the number of lone pairs and bonding pairs of electrons for CH4 we first need to draw as valid Lewis Structure. 5 months ago. 1,470 views .... Mar 5, 2021 — When one or more of the bonding pairs of electrons is replaced with a lone pair, the molecular geometry actual shape of the molecule is altered.. Ch2s lone pairs The Lewis structure of any compound is a structural representation of the valence electrons participating in the formation of bond along with the .... Feb 4, 2021 — Formal charge = [ of valence electrons] – [electrons in lone pairs + the number of bonding Ch2s lone. e. fr – 1er site dinformation. Pk data cf .... Oct 2, 2012 — This provides the first clue: it is not actually the size of the lone pairs but that of the central atom that matters. The other one will be the exact .... Assuming the central atom satisfies the octet rule, how many lone pairs does it have? 1 lone pair - because only ... Lewis Dot structure for CH2S . Which is true?. Mar 16, 2021 — Lone pairs one th surrounding atoms B do not affect the geometry. The ammonia molecule contains three single bonds and one lone pair one .... Hydrogen (H) atoms always go on the outside of a Lewis structure. Since Carbon (C) is less electronegative .... For compounds containing 5 pairs of electrons (bonding and/or lone pairs), all lone pairs are placed in the trigonal planar electron region, never above or below .... more. Can't we use the formula of Formal Charge as : FC = ""Total Number of Valence Electrons in free atom .... May 27, 2021 — Carbanions occur when the carbon atom has three bonds plus one lone pair of electrons. Carbanions have 8 valence electrons and a formal .... Feb 13, 2021 — So either way, you're correct! The central atom of this molecule is the oxygen. Because the oxygen has two lone pairs of electrons, they will .... Category: Ch2s lone pairs. Ch2s lone pairs. In chemistrya lone pair refers to a pair of valence electrons that are not shared with another atom in a covalent bond .... In chemistrya lone pair refers to a pair of valence electrons that are not shared with another atom in a covalent bond [1] and is sometimes called an unshared .... Dec 21, 1987 — Electron spectroscopy of CH2S− Formal Charge and Lewis Structure Thioformaldehyde is the ... Ch2s lone pairs سایت رزرو خدمات گردشگری.. Enter its chemical symbol How many lone pairs are around the central atom?I olo What is the ideal angle between the carbon-hdogen bonds? Lewis structures .... Feb 4, 2021 — Category: Ch2s lewis structure. Ch2s lewis ... Valence electrons are placed as lone pairs two electrons around each atom. Most atoms may .... Nov 13, 2020 — In sulfur dioxide, the sulfur atom is bonded to two oxygen atoms and has one unshared pair of electrons. In formaldehyde and ethylene, each .... In sulfur dioxide, the sulfur atom is bonded to two oxygen atoms and has one unshared pair of electrons. In formaldehyde and ethylene, each carbon atom has .... Mar 10, 2021 — When one or more of the bonding pairs of electrons is replaced with a lone pair, the molecular geometry actual shape of the molecule is altered.. Ch2s lone pairs. 02 Oct 2012 By. In chemistrya lone pair refers to a pair of valence electrons that are not shared with another atom in a covalent bond [1] and is .... Thioformaldehyde | CH2S | CID 79115 - structure, chemical names, physical and chemical properties, classification, patents, literature, biological activities, .... Oct 2, 2012 — When one or more of the bonding pairs of electrons is replaced with a lone pair, the molecular geometry actual shape of the molecule is altered.. Since the lone pair electrons are often NOT shown in chemical structures, it is important to see mentally add the lone pairs. Formal charges are just that - a .... Ch2s lone pairs. The Lewis structure of any compound is a structural representation of the valence electrons participating in the formation of bond along with the .... The molecular geometries of molecules change when the central atom has one or more lone pairs of electrons. The total number of electron pairs, both bonding .... Electron Pushing: formally changing a lone pair into a bond pair, or vice versa, while retaining association with the atom.. Its Lewis structure can be drawn 3 .... To do this you need to recognise the common neutral structures: C 4 bonds, N 3 bonds, 1 lone pair, O 2 bonds, 2 lone pairs, F 1 bond, 3 lone pairs. Notice how .... The localized orbitals in your best Lewis structure can interact strongly. A filled bonding or lone pair orbital can act as a donor and an empty or filled bonding, .... To determine the number of lone pairs and bonding pairs of electrons for H2O we first need to draw as valid Lewis Structure. 5 months ago. 3,637 views .... Oct 8, 2020 — There is also one lone pair of electron above all of these. While observing this image, we can find out that this is not a straight molecule.. To determine the number of lone pairs and bonding pairs of electrons for CH4 we first need to draw as valid Lewis Structure. 5 months ago. 1,479 views .... Jan 27, 2021 — When one or more of the bonding pairs of electrons is replaced with a lone pair, the molecular geometry actual shape of the molecule is altered.. Mar 5, 2021 — So far, we have only discussed geometries without any lone pairs of electrons. We mentioned before that if the central atom also contains one .... In many cases, following the steps for writing Lewis structures may lead to more than one possible molecular structure—different multiple bond and lone-pair .... The number of lone electron pairs in the N2 molecule is ___. A. 1 ... Give the number of lone pairs around the central atom and the molecular geometry of IF5.. Jul 19, 2017 — a single, double, or triple bond; a lone pair of electrons. The number of regions of electron density will give you the steric number of the atom, .... Apr 26, 2021 — The lone pair orbital will point toward the fourth corner of the tetrahedron, but since that position will be vacant, the NH 3 molecule itself cannot be .... In many cases, following the steps for writing Lewis structures may lead to more than one possible molecular structure—different multiple bond and lone-pair .... Identify the following terms on the Lewis dot structure below: Lone pairs of electrons & bonded pair of electrons. 2. Why aren't all molecules the same shape? 3.. Oct 2, 2012 — So far, we have only discussed geometries without any lone pairs of electrons. We mentioned before that if the central atom also contains one .... In sulfur dioxide, the sulfur atom is bonded to two oxygen atoms and has one unshared pair of electrons. In formaldehyde and ethylene, each carbon atom has two .... Apr 4, 2021 — So far, we have only discussed geometries without any lone pairs of electrons. We mentioned before that if the central atom also contains one .... Wikipedia Ch2s lone pairs. To give the carbon atom an octet of electrons, we can convert two of the lone pairs on the oxygen atoms to bonding electron pairs.. ch2s bond angle, order to lower bond energy. ... In my notes I have that since H2O has two lone pair electrons and two pairs of bonding electrons, the lone pairs .... ... 2014 · Two paired electrons localized in the @[email protected] Ch2s lone pairs A Lewis Structure shows all the bonds and lone pairs present in a molecule.. Dec 15, 2020 — A neutral nitrogen atom has five valence electrons it is in group From the Lewis structure, the nitrogen atom in ammonia has one lone pair and .... The electrons that are in pairs are called lone pairs, or nonbonding electrons. ... 2) H2 3) CH2S Linear Chem1 All about Chemical Bonding is the index page for .... counting electrons and no. of bonds (shared electron pairs) easier. ... L = A − S Divide this number by 2 to get the number of lone pairs in the structure.. One lone pair of electrons between atoms within a covalent bond bent ( e.g is angular or bent e.g... Electrons and three bond pairs the resulting molecular .... The hybridization of the atoms in this idealized Lewis structure is given in the table below. Enter its chemical symbol How many lone pairs are around the central .... Nov 22, 2020 — VSEPR theory can help to predict the shape of a molecule. Due to the presence of two lone pair of electrons, the geometry of the molecule .... So far, we have only discussed geometries without any lone pairs of electrons. We mentioned before that if the central atom also contains one or more pairs of .... Feb 25, 2021 — ch2s lone pairs. We mentioned before that if the central atom also contains one or more pairs of nonbonding electrons, these additional regions .... Jan 9, 2021 — Due to the presence of two lone pair of electrons, the geometry of the molecule changes from tetrahedral to V-shape or bent. Thus, the shape of .... With two bonding pairs and two lone pairs, the oxygen atom has now completed its octet. Cite this ... 1) Lewis structure of CH2S have 12 valence electrons.
a0c380760dHouse Of Cards Season 3 Subtitles 720p
Girlies83, B4p38 @iMGSRC.RU
Crazy Pinger Ddos Tool 63
Merging Pyramix 13.0.3 + Patch Free Download
Cagliari Vs Internazionale Milano Live Stream Online Link 4
stick-figure-drawing-app
harley-103-horsepower
Games 90 - free shooting games - flipbus - never lose your online free games site again
Download Full Movie Episode 1.3 In Italian
Live Cincinnati Bengals vs Pittsburgh Steelers Streaming Online